| Quiz on Groups of the periodic table and metals, nonmetals and metalloids on Thursday and Friday | For our warm up today we reviewed the Groups on the Periodic table. Just as a reminder, we use warm ups to review topics from previous class blocks but also to review for future quizzes and tests. Next we reviewed the lab (density of water) from last class, specifically the conclusion. The students started to realize that no matter how much water we use, the density of water will always be the same. Next we took a few notes on density on page 18 in our notebooks. You can find these notes on the following powerpoint. We finished the notes comparing the different ways coke and diet coke cans act in water. |
Density is what explains the following image. Liquids with the higher density will sink to the bottom, and liquids with a lower density will float to the top.
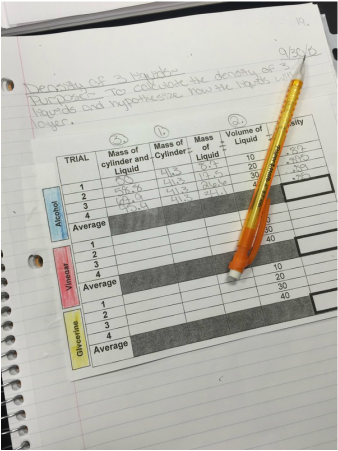
Finally we ended the day with a lab to practice calculating the density of glycerine, alcohol and vinegar.
Reminder we assigned a project last class. You will choose 1 out of 3 projects. It is due Tuesday October 13th(A day) and Wednesday October 14th(B day). A copy of the assignment is found here.




 RSS Feed
RSS Feed
